- Blog
- Ww2 navy waves patch
- Free neurotracker app
- Imvuksa texture cracked
- Mp3 old phone ringtone
- Adobe lightroom 6 serial numbers
- Free ntfs undelete
- Serious sam 1 mas resolucion pc
- Real rock riddim wav instrumental
- Free eye candy 4000 plugins download for paint shop pro
- Crimson court provisions
- Derivative of log formula
- Yu gi oh arc v
- Acronis true image 2014 reviews
- Threat of new entrants grocery industry
- Tabla beats ringtones free download
- Myatt app for windows
- What is driverdoc
- Train simulator 2019 japan
- Divinity original sin 2 leveling guide
- Av vcs diamond 9-5 full version
- Winners go home and fuck the prom queen quote
- Ford ids vs autoenginuity
- Beast battle simulator igg games
- Jonathan anderson neurotracker
- Divi dead psp iso espa-ol

Notice the fact that these functions are actually inverses of each other. The Henderson-Hasselbalch equation is useful in estimating the pH of buffer solutions as can be seen in the applications and examples page. Now, let’s look at how the derivatives for the logarithmic function are calculated. Hence it is very useful in fields of chemistry, biochemistry, and pharmacy. This is useful for certain reactions as well as important for absorption, distribution and excretion of drugs. The Henderson-Hasselbalch equation gives you information of the abundance of ionized and unionized fractions of molecules within a given solution. As we have seen above, the pK a gives an idea of the acidity and basicity of molecules, and is hence very useful in pharmaceutical, chemistry, biology and biochemistry fields.Ģ] If a molecule is placed in a solution of particular pH, what fraction of the molecule will be ionized vs unionized? The Henderson-Hasselbalch equation lets you calculate the pK a of a molecule using simple experimental protocols. The equation can basically tell you the following:ġ] How acidic or basic the molecule is? i.e. What Information the Henderson-Hasselbalch Equation Gives You?Ĭheck out the applications and examples of using Henderson-Hasselbalch equation. While, a strong base such as ammonia (NH 3) has a pK a of 38. For example, the pK a values for strong acids such as HCl can even go into the negative values with a pK a of -8. Any value higher than this pK a value increases basicity, while values which decrease from this value are increasing in acidity. The pK a value of acids and bases gives an idea of how strong the acids or bases are. These terms were made to judge whether a reaction would go forward or not. It must always be kept in mind that acidity and basicity are relative terms. PH = pK a – log (/) The pK a Scale for Acidity and Basicity Subtracting 14 on both sides and multiplying both sides by -1 we get the Henderson-Hasselbalch equation for bases in terms of pKa and pH: POH = pK b + log (/) may be rewritten as: Thus for bases, the Henderson-Hasselbalch equation: Thus, since pK a = -log K a and pK b = -log K b: The product of K a and K b is the dissociation constant of water: To derive the relationship for bases in terms of pH and pK a we must look at the relationship between pK a and pK b as well as the relationship between pH and pOH. However, in order to compare acidity with basicity the pK a of a base may also be calculated in order to keep a single scale of comparison.

Become a member to unlock this answer Create your account. Generally the term pK a can be used to measure the strength of an acid and pK b for bases. One type of function that has a nice derivative formula is a logarithmic function. POH = pK b + log (/) pK a And pH Can Be Used Universally For Both Acids and Bases Rearranging the terms we get the Henderson-Hasselbalch equation for bases: Since pK b = -log 10 K b and pOH = -log 10, therefore: Splitting the log terms into separate components gives you: Where and are the concentrations of the ionized form of the base, while is the concentration of the unionized form.
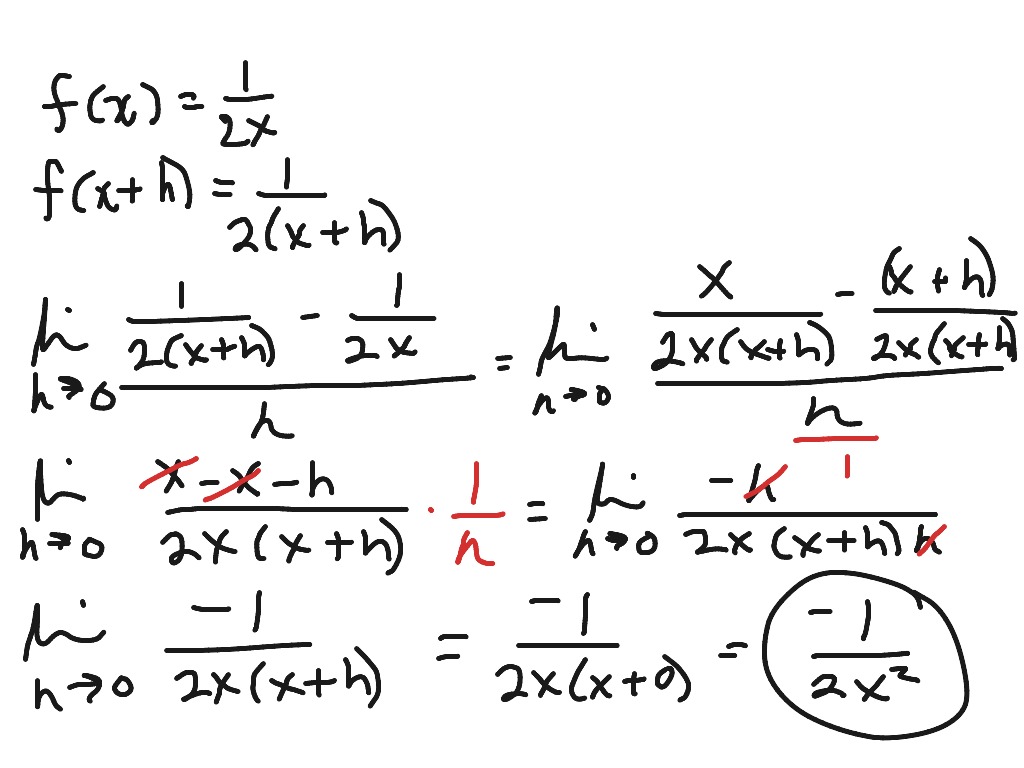
Examples of finding the derivative of lnx